You’ve probably noticed widespread TV advertisements lately for Cymbalta, Eli Lilly’s blockbuster antidepressant. However, these ads say nothing about depression. Sure, some of the actors may look a little depressed (the guy at right, from the Cymbalta web site, sure looks bummed), but the ads are instead promoting Cymbalta for the treatment of chronic musculoskeletal pain, an indication that Cymbalta received in August 2010, strengthening Cymbalta’s position as the “Swiss Army knife” of psychiatric meds. (I guess that makes Seroquel the “blunt hammer” of psych meds?)
You’ve probably noticed widespread TV advertisements lately for Cymbalta, Eli Lilly’s blockbuster antidepressant. However, these ads say nothing about depression. Sure, some of the actors may look a little depressed (the guy at right, from the Cymbalta web site, sure looks bummed), but the ads are instead promoting Cymbalta for the treatment of chronic musculoskeletal pain, an indication that Cymbalta received in August 2010, strengthening Cymbalta’s position as the “Swiss Army knife” of psychiatric meds. (I guess that makes Seroquel the “blunt hammer” of psych meds?)
Cymbalta (duloxetine) had already been approved for diabetic neuropathy and fibromyalgia, two other pain syndromes. It’s a “dual-action” agent, i.e., an inhibitor of the reuptake of serotonin and norepinephrine. Other SNRIs include Effexor, Pristiq, and Savella. Of these, only Savella has a pain [fibromyalgia] indication.
When you consider how common the complaint of “pain” is, this approval is a potential gold mine for Eli Lilly. Moreover, the vagueness of this complaint is also something they will likely capitalize upon. To be sure, there are distinct types of pain—e.g., neuropathic, visceral, musculoskeletal—and a proper pain workup can determine the exact nature of pain and guide the treatment accordingly. But in reality, overworked primary clinicians (not to mention psychiatrists, for whom hearing the word “pain” is often the extent of the physical exam) often hear the “pain” complaint and prescribe something the patient says they haven’t tried yet. Cymbalta is looking to capture part of that market.
 The analgesic mechanism of Cymbalta is (as with much in psychiatry) unknown. Some have argued it works by relieving the depression and anxiety experienced by patients in pain. It has also been proposed that it activates “descending” pathways from the brain, helping to dampen “ascending” pain signals from the body. It might also block NMDA receptors or sodium channels or enhance the body’s own endorphin system. (Click on the figure above for other potential mechanisms, from a recent article by Dharmshaktu et al., 2011.)
The analgesic mechanism of Cymbalta is (as with much in psychiatry) unknown. Some have argued it works by relieving the depression and anxiety experienced by patients in pain. It has also been proposed that it activates “descending” pathways from the brain, helping to dampen “ascending” pain signals from the body. It might also block NMDA receptors or sodium channels or enhance the body’s own endorphin system. (Click on the figure above for other potential mechanisms, from a recent article by Dharmshaktu et al., 2011.)
But the more important question is: does it work? There does seem to be some decent evidence for Cymbalta’s effect in fibromyalgia and diabetic neuropathy in several outcome measures, and in a variety of 12-week trials summarized in a recent Cochrane review.
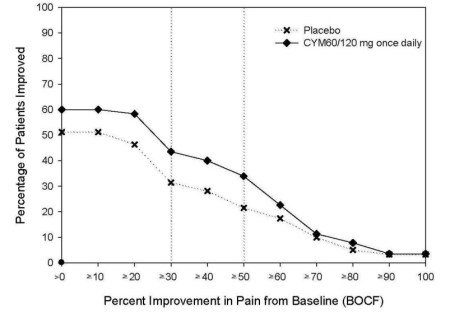 The evidence for musculoskeletal pain is less convincing. In order to obtain approval, Lilly performed two studies of Cymbalta in osteoarthritis (OA) and three studies in chronic low back pain (CLBP). All CLBP studies showed benefit in “24-hour pain severity” but only one of the OA studies showed improvement. The effects were not tremendous, even though they were statistically significant (see example above, click to enlarge). The FDA panel expressed concern “regarding the homogeneity of the study population and the heterogeneity of CLBP presenting to physicians in clinical practice.” In fact, the advisory committee’s enthusiasm for the expanded indication was somewhat muted:
The evidence for musculoskeletal pain is less convincing. In order to obtain approval, Lilly performed two studies of Cymbalta in osteoarthritis (OA) and three studies in chronic low back pain (CLBP). All CLBP studies showed benefit in “24-hour pain severity” but only one of the OA studies showed improvement. The effects were not tremendous, even though they were statistically significant (see example above, click to enlarge). The FDA panel expressed concern “regarding the homogeneity of the study population and the heterogeneity of CLBP presenting to physicians in clinical practice.” In fact, the advisory committee’s enthusiasm for the expanded indication was somewhat muted:
Even though the committee also complained of the “paucity of sound data regarding the pharmacological mechanisms of many analgesic drugs … and the paucity of sound data regarding the underlying pathophysiology,” they ultimately voted to approve Cymbalta for “as broad an indication as possible,” in order for “the well-informed prescriber [to] have the option of trying out an analgesic product approved for one painful condition in a patient with a similar painful condition.”
 Incidentally, they essentially ignored the equivocal results in the OA trials, reasoning instead that it was OK to “extrapolate the finding [of efficacy in CLBP] to other similar musculoskeletal conditions.”
Incidentally, they essentially ignored the equivocal results in the OA trials, reasoning instead that it was OK to “extrapolate the finding [of efficacy in CLBP] to other similar musculoskeletal conditions.”
In other words, it sounds like the FDA really wanted to get Cymbalta in the hands of more patients and more doctors.
As much as I dislike the practice of prescribing drugs simply because they’re available and they might work, the truth of the matter is, this is surely how Cymbalta will be used. (In reality, it explains a lot of what we do in psychiatry, unfortunately.) But pain is a complex entity. We have to be certain not to jump to conclusions—like we frequently do in psychiatry—when/if we see a “success story” with Cymbalta.
To the body, 60 mg of duloxetine is 60 mg of duloxetine, whether it’s being ingested for depression or for pain. If a patient’s fibromyalgia or low back pain is miraculously “cured” by Cymbalta, there’s no a priori reason to think that it’s doing anything different in that person than what it does in a depressed patient (even though that is entirely conceivable). The same mechanism might be involved in both.
The same can be said for some other medications with multiple indications. For example, we can’t necessarily posit alternate mechanisms for Abilify in a bipolar patient versus Abilify in a patient with schizophrenia. At roughly equivalent doses, its efficacy in the two conditions might be better explained by a biochemical similarity between the two conditions. (Or maybe everything really is bipolar! —sorry, my apologies to Hagop Akiskal.)
Or maybe the medication is not the important thing. Maybe the patient’s perceived need for the medication matters more than the medication itself, and 60 mg of duloxetine for pain truly is different from 60 mg duloxetine for depression. However, if our explanations rely on perceptions and not biology, we’re entering the territory of the placebo effect, in which case we’re better off skipping duloxetine (and its side effect profile and high cost), and just using an actual placebo.
 Bottom line: We tend to lock ourselves into what we think we know about the biology of the condition we’re treating, whether pain, depression, schizophrenia, ADHD, or whatever. When we have medications with multiple indications, we often infer that the medication must work differently in each condition. Unless the doses are radically different (e.g., doxepin for sleep vs depression), this isn’t necessarily true. In fact, it may be more parsimonious to say that disorders are more fundamentally alike than they are different, or that our drugs are being used for their placebo effect.
Bottom line: We tend to lock ourselves into what we think we know about the biology of the condition we’re treating, whether pain, depression, schizophrenia, ADHD, or whatever. When we have medications with multiple indications, we often infer that the medication must work differently in each condition. Unless the doses are radically different (e.g., doxepin for sleep vs depression), this isn’t necessarily true. In fact, it may be more parsimonious to say that disorders are more fundamentally alike than they are different, or that our drugs are being used for their placebo effect.
We can now add chronic pain to the long list of conditions responsive to psychoactive drugs. Perhaps it’s also time to start looking at pain disorders as variants of psychiatric disorders, or treating pain complaints as symptoms of mental disorders. Cymbalta’s foray into this field may be the first attempt to bridge this gap.
Addendum: I had started this article before reading the PNAS article on antidepressants and NSAIDs, which I blogged about earlier this week. If the article’s conclusion (namely, that antidepressants lose their efficacy when given with pain relievers) is correct, this could have implications for Cymbalta’s use in chronic pain. Since chronic pain patients will most likely be taking regular analgesic medications in addition to Cymbalta, the efficacy of Cymbalta might be diminished. It will be interesting to see how this plays out.




 Posted by stevebMD
Posted by stevebMD