 The future of psychiatric diagnosis and treatment lies in the discovery and development of “biomarkers” of pathological processes. A biomarker, as I’ve written before, is something that can be measured or quantified, usually from a biological specimen like a blood sample, which helps to diagnose a disease or predict response to a treatment.
The future of psychiatric diagnosis and treatment lies in the discovery and development of “biomarkers” of pathological processes. A biomarker, as I’ve written before, is something that can be measured or quantified, usually from a biological specimen like a blood sample, which helps to diagnose a disease or predict response to a treatment.
Biomarkers are the embodiment of the new “personalized medicine”: instead of wasting time talking to a patient, asking questions, and possibly drawing incorrect conclusions, the holy grail of a biomarker allows the clinician to order a simple blood test (or brain scan, or genotype) and make a decision about that specific patient’s case. But “holy grail” status is elusive, and a recent study from the Yale University Department of Psychiatry, published this month in the journal Biological Psychiatry, provides yet another example of a biomarker which is not quite there—at least not yet.
The Yale group, led by Rajita Sinha, PhD, were interested in the question, what makes newly-abstinent cocaine addicts relapse?, and set out to identify a biological marker for relapse potential. If such a biomarker exists, they argue, then it could not only tell us more about the biology of cocaine dependence, craving, and relapse, but it might also be used clinically, as a way to identify patients who might need more aggressive treatment or other measures to maintain their abstinence.
 The researchers chose BDNF, or brain-derived neurotrophic factor, as their biomarker. In studies of cocaine-dependent animals who are forced into prolonged abstinence, those animals show elevations in BDNF when exposed to a stressor; moreover, cocaine-seeking is associated with BDNF elevations, and BDNF injections can promote cocaine-seeking behavior in these same abstinent animals. In their recent study, Sinha’s group took 35 cocaine-dependent (human) patients and admitted them to the hospital for 4 weeks. After three weeks of NO cocaine, they measured blood levels of BDNF and compared these numbers to the levels measured in “healthy controls.” Then they followed all 35 cocaine users for the next 90 days to determine which of them would relapse during this three-month period.
The researchers chose BDNF, or brain-derived neurotrophic factor, as their biomarker. In studies of cocaine-dependent animals who are forced into prolonged abstinence, those animals show elevations in BDNF when exposed to a stressor; moreover, cocaine-seeking is associated with BDNF elevations, and BDNF injections can promote cocaine-seeking behavior in these same abstinent animals. In their recent study, Sinha’s group took 35 cocaine-dependent (human) patients and admitted them to the hospital for 4 weeks. After three weeks of NO cocaine, they measured blood levels of BDNF and compared these numbers to the levels measured in “healthy controls.” Then they followed all 35 cocaine users for the next 90 days to determine which of them would relapse during this three-month period.
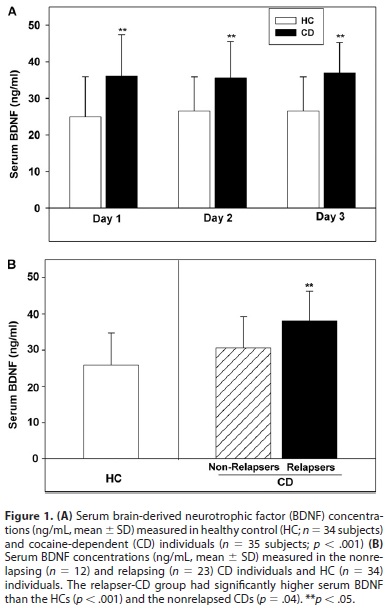
The results showed that the abstinent cocaine users generally had higher BDNF levels than the healthy controls (see figure below, A). However, when the researchers looked at the patients who relapsed on cocaine during the 3-month follow-up (n = 23), and compared them to those who stayed clean (n = 12), they found that the relapsers, on average, had higher BDNF levels than the non-relapsers (see figure, B). Their conclusion is that high levels of BDNF may predict relapse.
These results are intriguing, and Dr Sinha presented her findings at the California Society of Addiction Medicine (CSAM) annual conference last week. Audience members—all of whom treat drug and alcohol addiction—asked about how they might measure BDNF levels in their patients, and whether the same BDNF elevations might be found in dependence on other drugs.
But one question really got to what I think is the heart of the matter. Someone asked Dr Sinha: “Looking back at the 35 patients during their four weeks in the hospital, were there any characteristics that separated the high BDNF patients from those with low BDNF?” In other words, were there any behavioral or psychological features that might, in retrospect, be correlated with elevated BDNF? Dr Sinha responded, “The patients in the hospital who seemed to be experiencing the most stress or who seemed to be depressed had higher BDNF levels.”
Wait—you mean that the patients at high risk for relapse could be identified by talking to them? Dr Sinha’s answer shows why biomarkers have little place in clinical medicine, at least at this point. Sure, her group showed correlations of BDNF with relapse, but nowhere in their paper did they describe personal features of the patients (psychological test scores, psychiatric complaints, or even responses to a checklist of symptoms). So those who seemed “stressed or depressed” had higher BDNF levels, and—as one might predict—relapsed. Did this (clinical) observation really require a BDNF blood test?
 Dr Sinha’s results (and the results of others who study BDNF and addiction) make a strong case for the role of BDNF in relapse or in recovery from addiction. But as a clinical tool, not only is it not ready for prime time, but it distracts us from what really matters. Had Dr Sinha’s group spent four weeks interviewing, analyzing, or just plain talking with their 35 patients instead of simply drawing blood on day 21, they might have come up with some psychological measures which would be just as predictive of relapse—and, more importantly, which might help us develop truly “personalized” treatments that have nothing to do with BDNF or any biochemical feature.
Dr Sinha’s results (and the results of others who study BDNF and addiction) make a strong case for the role of BDNF in relapse or in recovery from addiction. But as a clinical tool, not only is it not ready for prime time, but it distracts us from what really matters. Had Dr Sinha’s group spent four weeks interviewing, analyzing, or just plain talking with their 35 patients instead of simply drawing blood on day 21, they might have come up with some psychological measures which would be just as predictive of relapse—and, more importantly, which might help us develop truly “personalized” treatments that have nothing to do with BDNF or any biochemical feature.
But I wouldn’t hold my breath. As Dr Sinha’s disclosures indicate, she is on the Scientific Advisory Board of Embera NeuroTherapeutics, a small biotech company working to develop a compound called EMB-001. EMB-001 is a combination of oxazapam (a benzodiazepine) and metyrapone. Metyrapone inhibits the synthesis of cortisol, the primary stress hormone in humans. Dr Sinha, therefore, is probably more interested in the stress responses of her patients (which would include BDNF and other stress-related proteins and hormones) than in whether they say they feel like using cocaine or not.
That’s not necessarily a bad thing. Science must proceed this way. If EMB-001 (or a treatment based on BDNF) turns out to be an effective therapy for addiction, it may save hundreds or thousands of lives. But until science gets to that point, we clinicians must always remember that our patients are not just lab values, blood samples, or brain scans. They are living, thinking, and speaking beings, and sometimes the best biomarker of all is our skilled assessment and deep understanding of the patient who comes to us for help.



 Posted by stevebMD
Posted by stevebMD 













